Hours and Hours of Study with no fun, is a bad idea for you, foreseeing the long run. Vedantu is the first choice of students aspiring to score full marks in their ICSE and CBSE Board exams or to crack any competitive exam like IIT JEE (Mains & Advanced), Kishore Vaigyanik Protsahan Yojana (KVPY), National Talent Search Exam (NTSE), International Math Olympiad (IMO), International English Olympiad (IEO). Interactive approach establishes a well-deserved academic connect between you and Master Teachers. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. 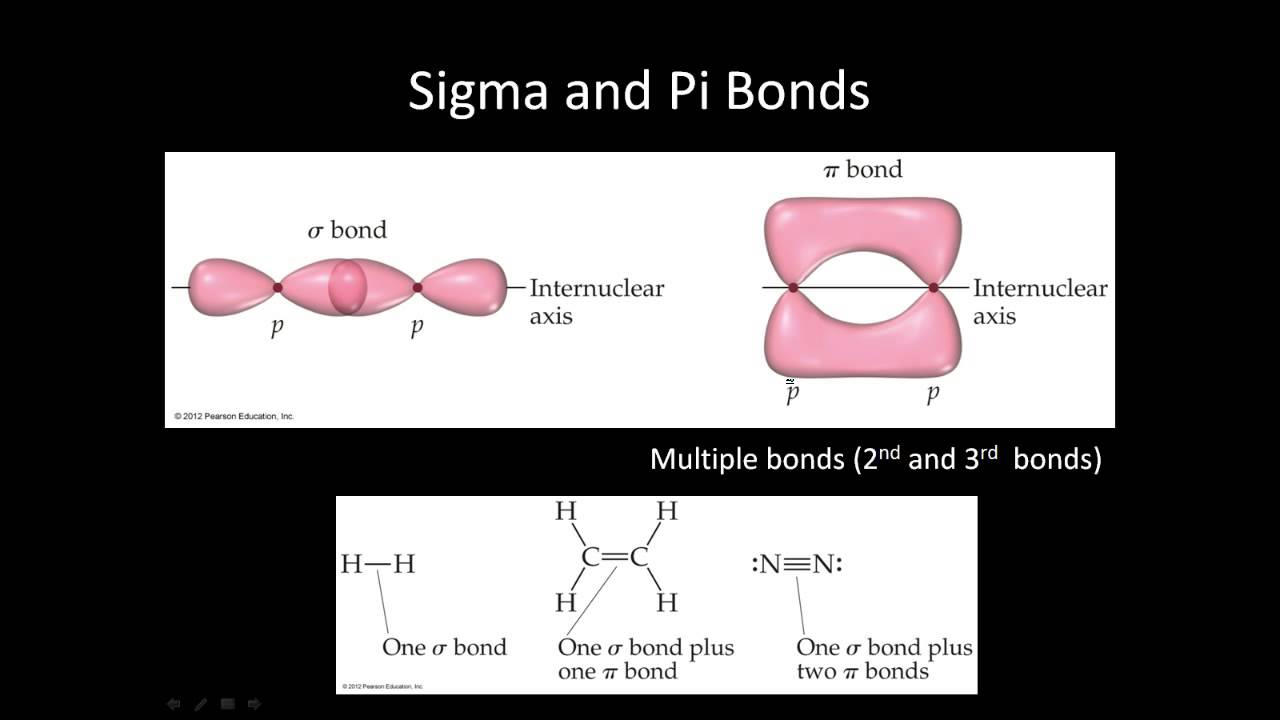
Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. The parallel orientation of the two p orbitals in adjacent atoms with proper sideways overlap results in the creation of a pi bond. S-s overlap, s-p overlap, and p-p overlap are the three most prevalent overlap conditions for forming sigma bonds. Sigma bonds are extremely strong covalent bonds, and sigma electrons are the electrons that participate in their creation. Pi bonds are created by the lateral overlap of two atomic orbitals, whereas sigma bonds are formed by the head-to-head overlapping of atomic orbitals. The overlapping of atomic orbitals is the main difference between these bonds. 
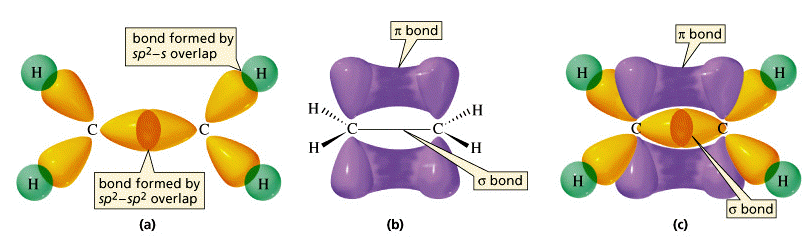
It is a truth that if two bonds are present, one must be sigma and the other a pi bond.Īlso, if there are three bonds, one of them will be a sigma bond, while the other two will be pi bonds. It is a proven truth that if two atoms form a single bond, the bond will be a sigma bond. Still, there are a few more key points to consider: Although the sigma and pi bond differences are complimentary, they are not convertible. With so many differences, it's clear that these two bonds are crucial. It doesn’t determine the shape of the molecule. It has cylindrical charge symmetry around the bond axis. Example- alkene and alkyne.Ītoms with sigma bonds are highly reactive.Ītoms with pi bonds are less reactive than atoms having sigma bonds only. In pi bond, overlapping orbitals are always pure orbitals only. In sigma bonds, overlapping orbitals can be pure orbitals, hybrid orbitals and one hybrid and one pure orbital. It is created by the linear overlap of the s-s, s-p, and p-p orbitals.Ĭovalent bond which is formed by the head on overlapping atomic orbitals is called sigma bond.Ĭovalent bond which is formed by lateral overlapping of the half-filled atomic orbitals of atoms is called pi bond. It is made up of atomic orbitals that overlap head-to-head.Īround the bond axis, it is cylindrically symmetrical.īecause of the maximal overlap, it is a significantly stronger bond. Formation of sigma bond is given below between the orbitals: We find sigma bonds in Alkanes, Alkenes, Alkynes. The strongest covalent bond which is formed by the head-on overlapping atomic orbitals is called the sigma bond. And also will discuss what is sigma and pi bond, the difference between the pi bond and sigma bond. Here in this article, we are going to discuss sigma and pi bonds which are covalent bonds only. Covalent bonds are those bonds that are formed by sharing of electrons between two atoms. Chemical bonds are classified into covalent bonds, coordinate bonds, ionic bonds, and hydrogen bonds. Chemical bonds are forces that keep atoms joined together.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
